
Pharmaceutical Metal Detection System for Quality Control for Tablets and Capsules
- Ultra High Sensitivity to all metals
- Enhanced product throughput rate
- Validation Solutions Available: Calibration Standards; Validation Manuals; Compliance Tools; Comprehensive Service & Support; On-line production data validation
- AISI 316L stainless steel construction
- Control Power Box available according to UL 508A and CSA-C22.2 No. 14-05
- 500 product data memories, selectable by local programming or network software
- 10.000 storable events
- High-definition graphic display
- Local programming: 16 keys, 3 with double function
- Quick Access key for fast programming of user parameters
- ATEX22 version (available on demand)
CEIA THS/PH21N Metal Detection Systems feature extremely high detection sensitivity towards contaminating metals, whether ferrous, non-ferrous or stainless steel, even when these are present in tiny quantities.
Options & Accesories
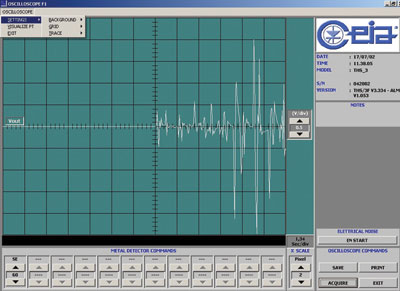 MDSCOPE is a software package specifically developed by CEIA in order to allow easy and complete testing of the functioning of a Metal Detector – without the use of special instrumentation – both during the setting up phase and on the occasion of periodic tests. Using a personal computer, MDSCOPE allows adjustment and monitoring of the Metal Detector, as well as the storage and printing of the data gathered.
MDSCOPE is a software package specifically developed by CEIA in order to allow easy and complete testing of the functioning of a Metal Detector – without the use of special instrumentation – both during the setting up phase and on the occasion of periodic tests. Using a personal computer, MDSCOPE allows adjustment and monitoring of the Metal Detector, as well as the storage and printing of the data gathered.


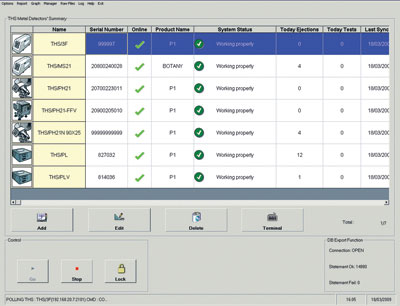


 Search, download and print certificates of compliance from the lot number engraved or printed on your test sample.
Search, download and print certificates of compliance from the lot number engraved or printed on your test sample.


